The bibliography of a clinical protocol serves to demonstrate that the protocol is built on a solid foundation of existing knowledge, ensuring that the study’s hypotheses and objectives are well-justified and scientifically valid.
The introduction of an article is meant to…introduce what has been done! But what structure can we give it?
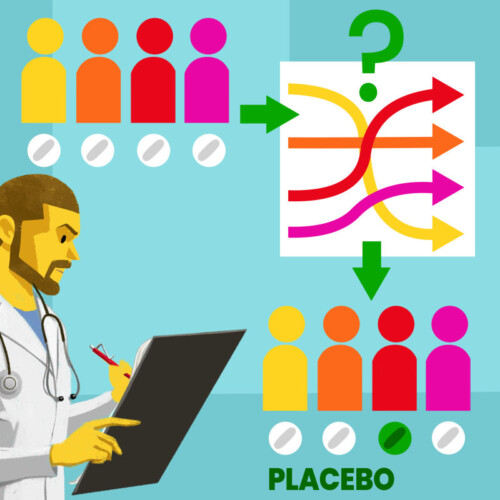
Randomization is a statistical procedure that, when applied in clinical studies, assigns participants to different treatment groups randomly.
News & Stories
Be informed and inspired by our weekly insights. An original perspective on regulatory affairs and quality assurance for medical devices and in vitro diagnostic medical devices, scientific communication and medical writing for the entire life science sector.
Subscribe to Clariscience Magazine
Be informed and inspired by our weekly insights. An original perspective on regulatory affairs and quality assurance for medical devices and in vitro diagnostic medical devices, scientific communication and medical writing for the entire life science sector.
The bibliography of a clinical protocol serves to demonstrate that the protocol is built on a solid foundation of existing…
The introduction of an article is meant to…introduce what has been done! But what structure can we give it?
Randomization is a statistical procedure that, when applied in clinical studies, assigns participants to different treatment groups randomly.
Inspection verification is a fundamental tool that companies can—and must—adopt to assess whether their Quality Management System (internal audit) or…
The risk management activity should be entrusted to a qualified team formed for this purpose, usually consisting of at least…
In any clinical study, researchers often encounter datasets with missing observations, commonly referred to as “missing data.”
According to the MDR Regulation, the technical documentation must be complete, well-structured, and contain sufficient information to demonstrate that the…
The data recording system aims to collect the results of all activities performed, all checks conducted, and the results obtained,…